Professor Gary Carvalho

- Name
- Prof. Gary Carvalho
- Position
- Research Professor of Molecular Ecology
- g.r.carvalho@bangor.ac.uk
- Phone
- +44(0)1248 382100
- Location
- School of Natural Sciences, Bangor University, Deiniol Road, Bangor, Gwynedd, LL57 2UW, UK
About
Using primarily DNA-based tools, my research is aimed at the elucidation of fundamental aspects of a species’ biology such as patterns of dispersal and gene flow, evolution of life histories and behaviour, response to environmental stress, and mechanisms of speciation, as well as the application of molecular tools to the management and conservation of exploited aquatic species from temperate, tropical and Antarctic marine and freshwater ecosystems.
Research includes the molecular analysis of population and species biodiversity of aquatic animals, with studies aimed at understanding the forces that shape genetic structure in the wild, and how such structure may influence adaptation, population persistence and distribution. Notable areas of activity include: the evolution and ecological significance of population differentiation, phylogeography and phylogenetics of aquatic taxa, the molecular analysis of past populations using PCR-based recovery of DNA (ancient DNA) from resting eggs and preserved material (e.g. fish otoliths and scales), DNA barcoding, traceability of fish and fish products, the evolutionary genetics of clonal animals, the evolution of adaptive traits using molecular, genomic and quantitative genetic analysis, and fisheries and conservation genetics of exploited fish in temperate, tropical and Antarctic waters.
In addition to current research, I contribute to various external bodies, as detailed in my CV, which include current roles as Vice-President of the Fisheries Society of the British Isles, Chair of the Expert ICES (International Council for Exploration of the Sea) Group on the Application of Genetics in Fisheries and Mariculture (WGAGFM), Vice-Chair of European Commission Panels (Marie Curie Actions and ITN), and Member of the Species Survival Commission (IUCN) Specialist Group in Conservation Genetics. I am currently Editor of the Proceedings of the Royal Society B, Fish and Fisheries, and members of the editorial boards of Molecular Ecology and Conservation Genetics. Gary Carvalho was awarded the Fisheries Society of the British Isles (FSBI) Beverton Medal in 2018, in recognition of ground-breaking research and life-long contribution to fish and fisheries science.
Recent significant areas of investigation include the use of environmental DNA (eDNA) for exploring the biodiversity of aquatic environments, and application of next generation sequencing for investigating microbiomes, especially in fishes. If you want to find out more, click on the research tab above.
The Bangor-based Molecular Ecology and Evolution at Bangor (MEEB) that I continue to coordinate now represents among the largest European groupings focusing on the molecular ecology and evolution of aquatic taxa. In addition to exploring fundamental issues in molecular ecology and evolution, MEEB contributed significantly to the 2014 UK Research Excellence Framework (REF) exercise, through both the inclusion of high impact outputs, and in the provision of an Impact Case Study focusing on wildlife forensics and traceability in marine fisheries. MEEB also contributes significantly at an international level, with members on the editorial teams of leading journals in the field, such as Proceedings of the Royal Society Lond. B, Molecular Ecology, Molecular Ecology Resources, Fish and Fisheries, and Conservation Genetics. It hosted an annual meeting of the UK Environmental DNA (eDNA) working group in 2015, preceded by a training workshop in eDNA for Indonesian colleagues. MEEB also coordinated a major international symposium on Fish, Genes and Genomes in July 2016 with leading key-note speakers such as Robin Waples, Craig Primmer, Louis Bernatchez, Dorte Bekkevold, Jenny Ovenden and John Casey. A special issue of the Journal of Fish Biology published the key note and selection of oral presentations (http://onlinelibrary.wiley.com/doi/10.1111/jfb.2016.89.issue-6/issuetoc). C
arvalho is also contributing to a EuroMarine workshop focusing on the Application of Genomics in Fisheries (https://goo.gl/g1cLun ), to be held in Portugal in May 2017. Currently, Gary Carvalho, while Chair of the ICES Expert Group on the Application of Genetics in Fisheries and Aquaculture, with other colleagues from the group, submitted a successful proposal to the ICES to host a new training course on “Genomics in Support of Fisheries and Aquaculture”, to take place in June 2018, at the Joint Research Centre, European Commission, Italy. For the first time, the ICES training portfolio offers a course on the design and application of genomics in management and conservation of fisheries and aquaculture. Our new course aims to narrow the science – policy divide by focusing on the need to render genomics more responsive and relevant to management needs, as supported by recent conceptual and technical advances. These will be placed within the context of salient policy frameworks. Further details can be found at: http://ices.dk/news-and-events/news-archive/news/Pages/Genomics-training-course.aspx.
More broadly, research in the School of Natural Sciences is ranked in the top 20 Universities in the UK (2014 REF). Across the University, three-quarters of Bangor’s research is either world-leading or internationally excellent.
CV
Education and honours
- FSBI Beverton Medal 2018, for ground-breaking research and life-long contribution to fish and fisheries science
- Fellowship of the Linnean Society (F.L.S.) (2000)
- Fellowship of the Society of Biology (F.S.B.) (2011)
- PhD. (Wales, 1985) – Ecological genetics of Daphnia
- M.Sc. (Wales, 1980) – Ecology
- B.Sc. (Class I, London, 1978) – Environmental Biology
Career
- 2009– Visiting Professor, University of Penang, Malaysia
- 2005–present Professor of Molecular Ecology, School of Biological Sciences/Environment Centre Wales, Bangor University
- 2006–2008 Deputy Head of School of Biological Sciences
- 2007- Chair, Environment Centre Wales, Management Board
- 1996–2004 Professor of Molecular Ecology, Dept. Biological Sciences, Hull University
- 1998–2004 Research Director, Department of Biological Sciences, Hull
- 1995–1996 Senior Lecturer in Marine Biology, University of Wales, Swansea
- 1990–1995 Lecturer in Marine Biology, Biological Sciences, University of Wales, Swansea
- 1988–1989 AFRC R.A. (University of Southampton), DNA fingerprinting of aphids
- 1986–1988 NERC Post-doctoral Fellowship (Univ. Wales, Bangor), population structure of isopods
- 1986–1987 Max-Planck-Society Research Fellowship (M.P.I. of Limnology, Germany), Clonal Structure of Daphnia
Editorial Work
- 1995–2000 Asst. Editor (Genetics and Evolution): Journal of Fish Biology
- 1996–2000 Editorial Board: Reviews in Fish Biology and Fisheries
- 1998–2003 Editorial Board: Heredity
- 2000 -present Editorial Board: Molecular Ecology
- 2000–2007 Associate Editor: Fish and Fisheries
- 2000 – present Editorial Board: Conservation Genetics
- 2003–2009 Editorial Board, Proceedings of Royal Society, London, B.
- 2007- present Editor, Fish and Fisheries
- 2011- present Editor, Proceedings of Royal Society, London, B.
- 2012–2013 Editorial Board, Biological Invasions
External Professional Activities
- 1998–present ICES Working Group on the Application of Genetics in Fisheries and Mariculture
- 2000–2007 NERC Steering Committee: Environmental Genomics Thematic Prog.
- 2003– NERC ad hoc Advisory Committee on Post-genomic Science and Proteomics
- 2003–2007 Management Group of Directors Working Group (CEFAS, DEFRA, DARDNI, FRS) on Cod Population Structure
- 2004–2008 Scientific Advisory Panel, Environment Agency
- 2004–2007 NERC Steering Committee, UK Molecular Genetics Facilities
- 2005–2009 Chair, European Coordinator of FISH-BOL (DNA Barcoding of Marine Fishes) and Steering Committee member
- 2005–2008 NERC Peer Review College
- 2005–2006 Vice-President of the Fisheries Society of the British Isles
- 2005–2006 Member of SEBI2010 Expert Group on Fishes (EEA/ECNC/UNEP). Streamlining European 2010 Biodiversity Indicators.
- 2006– Expert Evaluator Panel– Norwegian Research Council (Seas and the Oceans)
- 2007– Member Academy of Finland Biosiences Grant Review Panel– Ecology Panel
- 2007– Chair of NERC Moderating Panel A (Molecular Ecology & Evolution)
- 2008–present External Consultant on various programmes for FCT (Portuguese Research Council for Science and Technology)
- 2008– ERA-Net BiodivERsA Evaluation Committee (EU Commission)
- 2008–2011 Coordinator of EU FP 7 Consortium, FishPopTrace
- 2008: Member of Research Quality Review, University College Cork
- 2009–2011 FAO Expert Group on Fisheries Forensics
- 2009 Visiting Professor, University of Penang, Malaysia
- 2009–2010 Expert Group of Marine Biodiversity for IFREMER, French Ministry of the Environment
- 2010 Organising Committee,SNP III International Conference Seattle, Washington
- 2010 Organising Committee, ECBOL2:2nd European Conference on DNA Barcoding
- 2000–present Editorial Board: Molecular Ecology
- 2000–2007 Associate Editor: Fish and Fisheries
- 2008–present Editor: Fish and Fisheries
- 2000–present Editorial Board: Conservation Genetics
- 2003–2009 Editorial Board, Proceedings of Royal Society, London, B
- 2011–present Editor, Proceedings of Royal Society, London, B
- 2011 Guest Editor, Applications of SNP Genotyping in Non-model organisms, Molecular Ecology Resources
- 2012 Editorial Board, Biological Invasions
- 2012 – Founder Member – UK Biodiversity Science Committee – to represent the UK in the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystems (IPBES) and other International biodiversity initiatives
- 2012 – European Expert Group on Fish Stock Assessment (Joint Research Centre– “Assessment for All”– EC)
- 2012 – DEFRA Stakeholder Advisory Group on Fish Traceability
- 2012 – Appointment by Centre for Independent Experts, US – scientific advice on petition for two species of river herring (blueback and alewife) to be included under Endangered Species Act
- 2012–2014 – NERC Peer Review College and Panel Member, Natural Environment Research Council, UK
- 2013 – FAO Expert Advisor on Establishment of Advisory Group on Aquatic Genetic Resources, Bangkok
- 2013, 2014 – Chair, Academy of Finland Biosiences Grant Review Panel– Ecology Panel
- 2013 – Member of the Advisory Board and Task Force for the Natural Environment Research Council (NERC, UK) knowledge exchange program on sustainable fisheries & aquaculture
- 2013 – Co-Convenor of Congress of the European Society for Evolutionary Biology, special symposium: Genomic Islands: their role in adaptation and speciation (August, Lisbon)
- 2013 – Member and Chair of Review Panel, Belgian Research Action through Interdisciplinary Networks, Belgian Government, Belgian Science Policy Office, Brussels
- 2013, 2014 – Member of the European Commission Evaluation Panel for Marie-Curie Actions, Environment. Brussels
- 2013 – Independent External Advisor, Marine Stewardship Council, DNA testing and certification of sustainable fisheries
- 2014 – present – NERC Core Panel Member – Panel E and others.
- 2014–2017 – Chair, ICES Working Group on the Application of Genetics in Fisheries and Mariculture (WGAGFM)
- 2014 – Founder Member of Species Survival Commission (IUCN) Specialist Group in Conservation Genetics.
- 2015 – OECD Expert Group Member on the long-term potential of marine biotechnology
- 2015–2018 – Vice-President, Fisheries Society of the British Isles (FSBI)
- 2015–2018– Chair, FSBI Publications Committee
- 2016 – Co-Convenor Bioeconomics, Sociobiology and Other Mixes.The Advantage of Linking Disparate Data to Gain New Insights into the Exploitation of Marine Fish. Special Theme Session, World Fisheries Congress, South Korea (May, 2016)
- 2016 – Convener, Fisheries Society of the British Isles (FSBI) Annual International Symposium: Fish, Genes and Genomes: Contributions to Ecology, Evolution and Management. Bangor, UK, July 2016.
- 2016– present External assistant working with the Research Executive Agency– European Commission, Brussels
- 2018 Member of Peer Review Panel for Genome Canada, Genomics Solutions for Agriculture, Agri-Food, Fisheries and Aquaculture.
Outreach and Public Communication of Science
a. Communicating genetic principles to environmental managers
For over 2 decades, effort has been focused on communicating the value of molecular tools and genetic diversity as core components of environmental management and conservation of biodiversity, especially among exploited species. Targeted publications such as Special Issues of international fisheries journals (e.g. 1995, 2008) aimed to render genetic concepts and tools more accessible to fisheries managers and conservation biologists. Direct input has been facilitated by engagement in appropriate bodies charged with responsibility for managing natural resources (e.g. ICES Working Group on the Application of Genetics in Fisheries and Mariculture; Management Group of Directors Working Group (CEFAS, DEFRA, DARDNI, FRS) on Cod Population Structure; Member of SEBI2010 Expert Group on Fishes (EEA/ECNC/UNEP). Streamlining European 2010 Biodiversity Indicators; FAO Expert Group on Fisheries Forensics; Expert Group on Marine Biodiversity for IFREMER, French Ministry of the Environment, 2009–2010; Founder Member – UK Biodiversity Science Committee – to represent the UK in the Intergovernmental Science-Policy Platform on Biodiversity and Ecosystems (IPBES) and other International biodiversity initiatives; Expert Food and Agriculture Organization of the United Nations State Of World Consultation on Aquatic Genetic Resources, Bangkok).
In addition to various research projects focusing on fisheries genetics, a notable recent programme, FishPopTrace, provided an opportunity for directly influencing the revision of major policies (Revised EC Common Fisheries Policy). FishPopTrace, an EU FP7 project (2002–11), coordinated by Carvalho, aimed to generate forensic tools (primarily single nucleotide polymorphisms, SNPs), to deliver a breakthrough in the legal enforcement of regulatory policies within the fishing industry. The key quest was to trace fish back to localities or spawning populations: the target and unit of harvesting and spatially resolved policies. For the first time, by applying high differentiation SNP assays, in four commercial marine fish, on a pan-European scale, it was possible to correctly assign unknown individuals back to their source spawning populations with unprecedented accuracy (93–100%). Outputs demonstrated how application of gene-associated markers will likely revolutionize origin assignment and become highly valuable tools for fighting illegal fishing and mislabelling worldwide. Recognising the impact of FishPopTrace (e.g. in Annex 1of the Commission Regulation for establishing a control system to secure compliance with the rules of the Common Fisheries Policy (COM (2008)721 final), it was stated “It is interesting to note the innovative contribution of FishPopTrace …for implementation of modern technologies”. Such distinction, combined with the new regulations imposed by the EC (EC Regulation 1224/2009 (Art 13), requiring EU States to undertake pilot studies of novel traceability tools by 2013, the Department of Environment Food and Rural Affairs (DEFRA, UK Govt.), is tailoring FishPopTrace tools for use by the UK fishing industry. FishPopTrace, and direct partnership with the Joint Research Centre (the science wing of the European Commission), also provided opportunities for direct input to consultation exercises on the Revised Common Fisheries Policy, in particular the successful adoption of advanced technologies for use in traceability and legal enforcement of regulations covering importation and capture of fish and fish products. The FishPopTrace SNP tools are also now being extended to new marine fish species around Europe (sea bream, bass, turbot), to generate a DNA reference data base for tests of traceability of escapee fish back to fish farms, as well as monitoring impact on fitness of wild fish species.
In July 2013, Carvalho was appointed by the Marine Stewardship Council as an Independent External Advisor on DNA testing of fish and fish products in the context of certification of sustainable stocks. Recent public and Government concern over traceability of food products, in conjunction with sustainability targets, demands a concerted, transparent and robust application of forensic standard authentic tests.
b. Broader outreach activities
Societal interest in biodiversity and management of natural resources has secured regular opportunities for communicating scientific outputs to the general public directly and through the media. Most notably, since 2010, contributions have been made to the week-long Bangor Science Festival, through evening public lectures on topics such as sustainability and fisheries forensics, as well as engagement with school children to introduce them to the excitement and wonder of the natural world through illustrating the uses of DNA tools to environmental management. The recent award of a Royal Society Partnership Grant, for partnership with a comprehensive school on Anglesey (the first such award in North Wales), has provided an interactive opportunity to discuss our research on “climate change, Antarctica and ice fish” – all topics that generate curiosity and interest, especially how scientific evidence can be gathered to support notions of climate change. In 2013, we coordinated a day-long event (with over 500 public visitors), as part of an event “The Hidden World”, where we provided hands-on and interactive displays illustrating life in the Antarctic, the life history of ice fish, and how climate change will likely impact these fragile marine ecosystems. Pupils aged 13–15 from our local partnered School presented talks to the general public on their own work to date as part of the Partnership Grant.
There has also been much media interest in our work– including interviews on BBC Radio on advances in using genetic fingerprinting to monitor the invasion of pest aphids (BBC Radio Wales; various national newspapers); the use of allozymes to manage and conserve the Falkland Island Fishery (BBC North East, local news); and more recently the generation of DNA-based tools for use in tackling illegal fishing and eco-certification of fish and fish products: (BBC World Service; Science In Action: http://www.bbc.co.uk/programmes/p00s1t04); Radio 4, BBC Farming Today; episode of the BBC Science Programme, in May 2013, Bang Goes the Theory, on the use of DNA forensics in traceability of fish products; NERC podcast on illegal fishing; http://planetearth.nerc.ac.uk/multimedia/story.aspx?id=452), as well an numerous regional and national newspapers.
Research
I arrived at Bangor University in January 2005, having moved from the Molecular and Evolutionary Ecology Group at Hull University. My initial remit was to expand in particular, studies on the molecular ecology of aquatic animals, including the use of molecular markers in fish and fisheries genetics. Although most research projects incorporate DNA-based tools, my research is aimed at the elucidation of fundamental aspects of a species’ biology such as patterns of dispersal and gene flow, evolution of life histories and behaviour, response to environmental stress, and mechanisms of speciation, as well as the application of molecular tools to the management and conservation of exploited aquatic species from temperate, tropical and Antarctic marine and freshwater ecosystems. In addition to my existing areas of interest, my new position at Bangor provides additional opportunities to develop studies on functional aspects of adaptive variation through genomic analysis, links between dispersal, gene flow and physical oceanography in the sea, and the use of high throughout sequencing in DNA barcoding.
The Bangor-based Molecular Ecology and Evolution at Bangor (MEEB) that I continue to coordinate now represents among the largest European groupings focusing on the molecular ecology and evolution of aquatic taxa, which in 2013 will be extended further with the appointment of two new lecturers. Martin Taylor, who was a founder member of MEEB since 2005, on 1 January 2013 took up a new post as Senior Lecturer in Molecular Ecology at the University of East Anglia (martin.taylor@uea.ac.uk). The two new lecturers are: Dr Mark de Bruyn, who has been based in MEEB since his Marie-Curie Fellowship in 2009, and Dr Michael Knapp, former Research Fellow in the Allan Wilson Centre for Molecular Ecology and Evolution Department of Anatomy University of Otago. The new appointments signal a significant expansion in the strength and scope of research and training activities of MEEB, especially in the area of ancient DNA to explore the impacts of long-term environmental change on biodiversity, and to reveal novel insights into the demographic history of extinct species, especially birds and mammals.
The purpose-built suite of molecular ecology laboratories in the Environment Centre Wales (ECW) provides a fully integrated and dedicated platform for the molecular analysis of biodiversity in wild populations. In addition to on-going work on fundamental evolutionary processes more opportunities are available for engagement with environmental agencies through links with the Centre for Ecology and Hydrology (NERC), Environment Agency (UK), Forestry Commission and Countryside Council for Wales, all of which have representation within ECW. Additionally, we have string and productive links with the NERC Molecular Genetic Facility in Liverpool, providing access to training and facilities for analysis of gene expression with microarrays, ultra-sequencing (and digital transcriptomics) and bioinformatics. Such additional facilities dove-tail with recent key developments within MEEB.
Below I summarise some current key areas of interest:
Molecular analysis of population structure

Molecular markers can be used to examine the relative roles of various microevolutionary forces on the levels and significance of genetic differentiation among populations. For example, one current project, in conjunction with the British Antarctic Survey and University of Hull, aims to disentangle the relative roles of life history variation and hydrographic processes on dispersal and gene flow in Antarctic fishes. It is becoming increasingly apparent that despite high dispersal capacity, some marine species continue to exhibit significant population structuring, occasionally on fine geographic scales. We are also examining, in collaboration with the University of Aveiro, links between hydrographic variability and larval dispersal and recruitment in the shore crab Carcinus maenas (see Carla Domingues, MEEB projects). In addition to exploring the role of hydrography in dispersal, we aim to assess methods for population assignment, and in conjunction with fine-scale genotypic monitoring, the role of selective mortality in the early stages of population recruitment.
Use of ancient DNA to study long-term environmental change
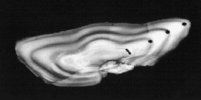 The availability of PCR enables recovery of DNA from natural (e.g. resting eggs of zooplankton: e.g. rotifers) and archived (e.g. fish scales and otoliths: e.g. cod) material to examine long-term changes in genetic structure. Using such approaches it is possible to examine the impact of natural and man-made environmental change on population and community structure. In collaboration with CEFAS (see Niklas Tysklind, MEEB projects) we are currently developing robust and sensitive methods for recovering DNA from archived histological samples of the flatfish, dab, Limanda limanda, as a basis for comparing genetic structuring in contemporary and past populations.
The availability of PCR enables recovery of DNA from natural (e.g. resting eggs of zooplankton: e.g. rotifers) and archived (e.g. fish scales and otoliths: e.g. cod) material to examine long-term changes in genetic structure. Using such approaches it is possible to examine the impact of natural and man-made environmental change on population and community structure. In collaboration with CEFAS (see Niklas Tysklind, MEEB projects) we are currently developing robust and sensitive methods for recovering DNA from archived histological samples of the flatfish, dab, Limanda limanda, as a basis for comparing genetic structuring in contemporary and past populations.
Evolution of adaptive variation

A core approach in molecular ecology has been to utilise neutral molecular markers to generate inferences about adaptive variation. Other approaches include either direct analysis of gene function or the use of quantitative genetic approaches to identify quantitative trait loci or to estimate heritabilities in biological traits. In conjunction with colleagues at the University of Hull we have for example: (1) examined geographic variation and performance under semi-controlled conditions of a key gene (Pantophysin I) associated with growth rate variation, (2) examined adaptive variation in freshwater bullhead using a combination of neutral and quantitative traits to assess heritabilities in relation to habitat variation.
Recently we have enhanced our approaches to exploring adaptive variation in the wild by encompassing transcriptomics to explore the impact of environmental stress on gene expression and associated adaptive phenotypes. Systems include Nucella lapillus and differential gene expression in response to environmental changes (natural and man-made), including the impact of contaminants on sex reversal (“imposex”) (See Sonia Pascoal, MEEB projects), and impact of environmental stress on gene expression in marine nematodes (see Simon Creer and Vera Fonseca, MEEB projects). We are the first to generate a transcriptome of a marine gastropod, and in early 2013, one of the major outcomes of the work will be published in Molecular Ecology, alongside a Perspective to explore its wider contribution to the field of genomics in non-model species. The paper: “Transcriptomics and in vivo tests reveal novel mechanisms underlying endocrine disruption in an ecological sentinel, Nucella lapillus”, provides novel insights into the control of imposex, and importantly, yields evidence for a common signalling pathway between invertebrate and vertebrate species that has previously been overlooked in the study of endocrine disruption. With funding from the NERC, and in conjunction with the Liverpool Microarray Facility, we are utilizing microarrays to explore genetic changes associated with phenotypic shifts associated with anadromy in brown trout, Salmo trutta (see Martin Taylor, MEEB).
Fisheries Genetics and Conservation
We utilise molecular genetic markers to examine the stock structure of commercial fishes, as well as examining demographic processes that may affect response to harvesting and environmental change. In particular we have utilised archived scales and otiliths to explore changes in genetic diversity and structure in relation to over-exploitation, and have demonstrated not only rapid short-term genetic change in North Sea cod during periods of population decline, but also a loss of genetic diversity following population collapse in New Zealand snapper. In conjunction with the Environment Agency, UK, our work now extends to examining population structure and biodiversity of salmonid fishes.
 Recent funding from the NERC (2008) has been obtained to examine the population structure of cod in UK coastal waters as part of the NERC-DEFRA-FRS-AFBI initiative, Sustainable Marine Bioresources. Many global marine fisheries have collapsed, or are at record low levels of abundance. Continuing exploitation and the uncertain impacts of climate change are adding further pressures on fish resources. New strategies are therefore required to assist in our management and conservation efforts. One such development will be to incorporate the extent and dynamics of spatially-associated biological differences that exist among fish stocks into stock assessment. Such information is important since most exploited fish species comprise assemblages of individuals that differ in their vital rates of growth, reproduction, migratory tendency and mortality. It therefore becomes desirable, for example, to match the level of fishing intensity to the projected rate of replenishment following harvesting, thereby reducing the probability that individual stocks will become extinct. Conserving such biological differences among stocks is also important in the maintenance of genetic diversity in wild fish populations, so endowing them with greater evolutionary potential for adapting to changes in the environment. A critical component of such work involves the testing of hypotheses relating to the relative contributions of such factors as dispersal of eggs and larvae, and behaviour of juveniles and adults in maintaining such biological differences. By integrating research efforts across each of the three major UK fisheries agencies (FRS, CEFAS, AFBI) with partners in UK Universities, a NERC Institute and an international cod expert from Denmark, we will for the first time examine the extent, patterns and stability of cod (Gadus morhua) population structuring throughout UK waters.
Recent funding from the NERC (2008) has been obtained to examine the population structure of cod in UK coastal waters as part of the NERC-DEFRA-FRS-AFBI initiative, Sustainable Marine Bioresources. Many global marine fisheries have collapsed, or are at record low levels of abundance. Continuing exploitation and the uncertain impacts of climate change are adding further pressures on fish resources. New strategies are therefore required to assist in our management and conservation efforts. One such development will be to incorporate the extent and dynamics of spatially-associated biological differences that exist among fish stocks into stock assessment. Such information is important since most exploited fish species comprise assemblages of individuals that differ in their vital rates of growth, reproduction, migratory tendency and mortality. It therefore becomes desirable, for example, to match the level of fishing intensity to the projected rate of replenishment following harvesting, thereby reducing the probability that individual stocks will become extinct. Conserving such biological differences among stocks is also important in the maintenance of genetic diversity in wild fish populations, so endowing them with greater evolutionary potential for adapting to changes in the environment. A critical component of such work involves the testing of hypotheses relating to the relative contributions of such factors as dispersal of eggs and larvae, and behaviour of juveniles and adults in maintaining such biological differences. By integrating research efforts across each of the three major UK fisheries agencies (FRS, CEFAS, AFBI) with partners in UK Universities, a NERC Institute and an international cod expert from Denmark, we will for the first time examine the extent, patterns and stability of cod (Gadus morhua) population structuring throughout UK waters.
We will employ existing (microsatellites) and new (single nucleotide polymorphisms) genetic markers to assess the extent to which individuals from different spawning groups interbreed, and will compare these indirect methods of dispersal with direct measures of fish movement taken from chemical analysis of ear-bones (”otoliths”) and individuals that have been electronically “tagged”. We will then input these data to tailored cod population models that will test the likely effects of young-stage and adult dispersal on observed patterns of stock separation, as well as simulating the likely consequences of spatially-based differences in stocks on such things as fishing effort, stock recovery and the design of marine protected areas. The proposal represents the first opportunity for UK Universities and all three UK Fisheries agencies to work together on a problem that has to take account of the interdependence of fish stocks across large regions of UK and adjoining waters. The proposal will generate new genetic estimates of stock separation, especially in areas of uncertainty such as the Celtic and Irish Seas and the Southern North Sea and English Channel, as well as the provision of new theoretical tools (population models) than can be used to forecast the impact of continued fishing pressure and environmental change on cod populations. Moreover, the tools will provide information on the appropriate spatial scale and distribution of marine protected areas and the probable rates of stock recovery in a species that is now formally endangered and listed in the IUCN Red List.
Carvalho (PI), Creer (Bangor co-I) and Taylor (recently moved to University of East Anglia (UEA)-co-I) were recently (2012) awarded a new EU FP7 project, AquaTrace, in partnership with a large consortium of European workers who aim to develop novel tools to explore the impact of aquaculture on native marine fish populations. The Bangor-UEA component of the work will focus on the phenotypic and genetic basis of fitness shifts associated with introgression between wild and captive-bred Atlantic salmon. The latter work will be carried out in conjunction with colleagues at the Bergen Marine Labaoratory, and will utilise state-of-the-art common garden facilities to explore fitness consequences and to map quantitative trait loci. Bangor will receive a PhD studentship that will be advertised early in 2013.
 A PhD project completed in 2011 (see Serinde Van Wijk, MEEB projects) examined the genetic basis of fisheries-induced evolution, using the Trinidadian guppy, Poecilia reticulata, as a model system. Size-selective harvesting can induce rapid phenotypic changes, such as age and size of maturation, in exploited fish populations. Despite the increasing incidence of such shifts, the relative contributions of genetic and environmental factors remain uncertain and are much-debated. Outputs from the study will appear early in 2013 in the journal, Frontiers in Ecology and the Environment, and demonstrate a significant genetic component to fisheries-induced evolution in an experimental system, with major implications for the sustainability of exploited populations, as well as impacts on size-structured communities and ecosystem processes, prompting a reconsideration of adaptation to, and recovery from, harvesting and predation. The work is now being extended by a PhD studentship awarded to Hazel Perry who will examine in more detail the genomic shifts associated with fisheries induced evolution in the model guppy system.
A PhD project completed in 2011 (see Serinde Van Wijk, MEEB projects) examined the genetic basis of fisheries-induced evolution, using the Trinidadian guppy, Poecilia reticulata, as a model system. Size-selective harvesting can induce rapid phenotypic changes, such as age and size of maturation, in exploited fish populations. Despite the increasing incidence of such shifts, the relative contributions of genetic and environmental factors remain uncertain and are much-debated. Outputs from the study will appear early in 2013 in the journal, Frontiers in Ecology and the Environment, and demonstrate a significant genetic component to fisheries-induced evolution in an experimental system, with major implications for the sustainability of exploited populations, as well as impacts on size-structured communities and ecosystem processes, prompting a reconsideration of adaptation to, and recovery from, harvesting and predation. The work is now being extended by a PhD studentship awarded to Hazel Perry who will examine in more detail the genomic shifts associated with fisheries induced evolution in the model guppy system.
Traceability of fish and fisheries forensics

MEEB was awarded (2008–2011) a major EU grant (FishPopTrace) to examine the traceability of fish and fish products in European waters, with focus on herring, cod, hake and sole. The 15 partner consortium is coordinated by Gary Carvalho and MEEB, and aims to develop a forensic framework for legal enforcement of policies aimed at reducing the amount of Illegal, Unreported and unregulated fishing (IUU). There is an increasing requirement for traceability of fish and fish products, both for consumer protection and for regulatory enforcement. For example, in the UK, the Marine Stewardship Council encourages consumers to eat particular landings of cod that are taken from ‘stocks maintained within safe limits’. A traceability system based on regional stocks is necessary to preclude fraudulent allocations. The underlying rationale of FishPopTrace is to assess and address challenges arising from the development of traceability tools within a forensic framework for four judiciously chosen target species: cod (Gadus morhua), hake (Merluccius merluccius), herring (Clupea harengus) and sole (Solea solea). While current information on levels of population structuring in traits such as life histories, morphometrics, genetics and physiology will be used to inform sample choice, new data will be restricted to markers at two levels: (1) Routine screening: selection of markers that exhibit maximal discriminatory power to identify populations, though with discrete and controlled variance enabling validation (single nucleotide polymorphisms (SNPs) and otolith microchemistry and morphometrics). Data from DNA-based methods provide a mechanism for traceability throughout the food supply chain (“fish to fork”) and indicate discrete spawning populations, whereas otoliths provide an independent on-board traceability system of fish provenance. (2) Testing of novel tools: additional tools will be tested on a selection of populations to assess validity and potential for traceability and validation, including fatty acid analysis, proteomics, gene expression analysis and the generation of high-throughput microarray platforms for SNP genotyping. Thus, FishPopTrace will provide information that relates to geography (“population tag”), as well as providing regional signatures that indicate biological differentiation in relation to spawning identity. Specifically, the consortium brings together recent and current expertise in fish traceability projects (Fish and Chips (GOCE-CT-2003–505491), FishTrace (QLRI-CT-2002–02755, FISH-BOL, Consortium for the Barcode of Life) to address several inter-related objectives:
1.1.2.1 To integrate recent and on-going data from European fish species traceability projects, and to generate a single compatible data base and tissue archive managed by the Joint Research Centre of the European Commission. The outputs will comprise a new data base and associated web links with access to recently generated data on fish species and population identity, together with an archive of associated tissue samples from external and consortium outputs.
1.1.2.2 To examine single nucleotide polymorphisms (SNPs) and otolith microchemistry and morphometrics in widely distributed populations of cod, hake, sole and herring, as tools for discriminating biologically differentiated populations and as a basis for traceability. Outputs will comprise population-level signatures associated with fish origins in early life and representative spawning groups.
1.1.2.3 To undertake validation of traceability tools in relation to end-user technology. Outputs will produce Standard Operating Procedures (SOPs) to allow transfer of technologies to other laboratories throughout EU member states.
1.1.2.4 To develop a population monitoring system based on otolith and genetic data that will assess population stability in a temporal and spatial framework. For each species, alternative parameters will be identified as indicators of population stability, and parameters will be validated using a combination of archived data and tissue samples.
1.1.2.5 To test the utility of additional novel traceability systems (fatty acid profiles, proteomics, gene expression, microarray platform for SNP genotyping). Outputs will comprise an assessment of the utility of additional novel approaches to traceability and population monitoring through estimating the within and among-population components of biological differentiation and population signatures. Additionally, a chip-based platform will be generated to undertake high-throughput SNP genotyping.
1.1.2.6 To facilitate technology transfer in relation to enforcement and conservation policies of the CFP and associated socio-economic consequences. Central elements of the output will be the development and evaluation of end-user tools, a Cost Benefit Analysis and a final report setting FishPopTrace in the context of the CFP.
A major output from the project was published in 2102 in Nature Communications. By applying high differentiation single nucleotide polymorphism assays, in four commercial marine fish, on a pan-European scale, we find 93–100% of individuals could be correctly assigned to origin in policy-driven case studies. We show how case-targeted single nucleotide polymorphism assays can be created and forensically validated, using a centrally maintained and publicly available database. Our results demonstrate how application of gene-associated markers will likely revolutionize origin assignment and become highly valuable tools for fighting illegal fishing and mislabelling worldwide.
Molecular Identification, DNA Barcoding and Phylochips

The use of short, standardised short DNA sequences – a “DNA barcode” has become a valuable addition to the taxonomic toolbox to validate species identity, as well as to identify unknown species, cryptic life history stages, etc. The Consortium for the Barcode of Life (CBOL) launched a new initiative in 2005 to DNA barcode all fishes- “FISH-BOL”, which will generate a global database for biologists and interested naturalists. In MEEB, I had the pleasure of acting as founder Chair of the European Regional working group for FishBol between 2005–2011. A past Marie-Curie Fellow, Dr Filipe Costa, is now the European Regional Chair, and continues to coordinate sample collection and data acquisition. Our DNA barcoding of marine fish was recently expanded to include novel data on a hot-spot of biodiversity – the Indo-Malay Archipelago. Some of the findings from a recent PhD programme with a local Malaysian student based in MEEB, Tun Nurul Aimi Mat Jaafar, was published in 2012 in PLoS ONE, and represents the first comprehensive analysis of cryptic species in a large ocmmercially important group, the Carangidae. Other DNA barcoding outputs have included the Crustacea, esp. Decapoda, from the completed 2012 PhD thesis of a MEEB-Portuguese FCT student, Joana Matzen, generating several papers detailed in the Carvalho publication list.
Phylochips are also being developed with funding from the NERC and in conjunction with the Liverpool Microarray Facility, to examine biodiversity of marine nematode communities (see Simon Creer, MEEB projects).

Selection of current research projects underway include:
- DNA barcoding of marine fishes (various sources);
- Population structure and traceability of marine fishes- development of novel tools (single nucleotide polymorphisms) to explore population diversity and traceability of fish and fish products in the food supply chain to tackle illegal fishing and promote consumer protection (various sources);
- Fisheries-induced evolution (Bangor University) – to assess the genetic and genomic basis of phenotypic shifts in size and age at maturation in exploited fish populations;
- The effects of Southern Ocean Warming on the development and population connectivity of Antarctic Ice fish (NERC);
- Use of metabarcoding and environmental DNA (eDNA) for biomonitoring of UK freshwaters (Knowledge Economy Skills Scholarships with Environment Agency, UK);
- The evolution of polyploidy and significance of gene duplication in tropical catfish (NERC);
- Development of tools for assessing the genetic impact of farmed fish on native fish populations (European Commission);
- Assessing the fitness consequences of introgression between captive and wild Atlantic salmon (European Commission);
- Conservation genetics and adaptive diversity of endangered manta rays (NERC)
- Meta-analysis of SNP diversity in globally distributed Atlantic cod (Gadus morhua) populations (Bangor University);
- Long-term changes in the biodiversity of moth comunities (Bangor University);
- Use of eDNA for monitoring biodiversity of freshwater communities in Malaysia (NERC).
- Reconstructing past demographic histories in relation to environmental change using ancient DNA (aDNA).
Publications
2024
- PublishedAn integrated spatio-temporal view of riverine biodiversity using environmental DNA metabarcoding
Perry, W. B., Seymour, M., Orsini, L., Jâms, I. B., Milner, N., Edwards, F., Harvey, R., de Bruyn, M., Bista, I., Walsh, K., Emmett, B., Blackman, R., Altermatt, F., Lawson Handley, L., Mächler, E., Deiner, K., Bik, H. M., Carvalho, G., Colbourne, J., Cosby, B. J., Durance, I. & Creer, S., 23 May 2024, In: Nature Communications. 15, 1, p. 4372
Research output: Contribution to journal › Article › peer-review
2023
- E-pub ahead of printComparative population genomics of manta rays has global implications for management
Humble, E., Hosegood, J., Carvalho, G., De Bruyn, M., Creer, S., Stevens, G. M. W., Armstrong, A., Bonfil, R., Deakos, M., Fernando, D., Froman, N., Peel, L. R., Pollett, S., Ponzo, A., Stewart, J. D., Wintner, S. & Ogden, R., 23 Nov 2023, (E-pub ahead of print) In: Molecular Ecology.
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental DNA metabarcoding for fish diversity assessment in a macrotidal estuary: A comparison with established fish survey methods
Gibson, T., Carvalho, G., Ellison, A., Gargiulo, E., Hatton-Ellis, T., Handley, L. L., Mariani, S., Collins, R. A., Sellers, G., Distaso, M., Zampieri, C. & Creer, S., 1 Nov 2023, In: Estuarine, Coastal and Shelf Science. 294, 108522.
Research output: Contribution to journal › Article › peer-review
2022
- PublishedCurrent and future genetic technologies for fisheries and aquaculture: implications for the work of FAO
Friedman, KJ., Bartley, DM., Rodríguez-Ezpeleta, N., Mair, GC., Ban, N., Beveridge, M., Carolsfeld, J., Carvalho, G., Cowx, I., Dean, G., Glazov, E., Leber, K., Loftus, R., Martinsohn, J., Olesen, I., Soto, E., Van Eenennaam, A. L. & Vigar, JR., 2022. 56 p.
Research output: Contribution to conference › Paper › peer-review - PublishedManaging human-mediated range shifts: understanding spatial, temporal and genetic variation in marine non-native species
Holman, L. E., Parker-Nance, S., de Bruyn, M., Creer, S., Carvalho, G. & Rius, M., 14 Mar 2022, In: Philosophical Transactions of The Royal Society B: Biological Sciences. 377, 1846, 20210025.
Research output: Contribution to journal › Article › peer-review
2021
- PublishedAnimals, protists and bacteria share marine biogeographic patterns
Holman, L. E., De Bruyn, M., Creer, S., Carvalho, G., Robidart, J. & Rius, M., 1 Jun 2021, In: Nature Ecology and Evolution. 5, 6, p. 738–746
Research output: Contribution to journal › Article › peer-review - PublishedDomestication-induced reduction in eye size revealed in multiple common garden experiments: The case of Atlantic salmon (Salmo salar L.)
Perry, W. B., Kaufmann, J., Solberg, M. F., Brodie, C., Medina, A. M. C., Pillay, K., Egerton, A., Harvey, A., Phillips, K. P., Coughlan, J., Egan, F., Grealis, R., Hutton, S., Leseur, F., Ryan, S., Poole, R., Rogan, G., Ryder, E., Schaal, P., Waters, C., Wynne, R., Taylor, M., Prodohl, P., Creer, S., Llewellyn, M., McGinnity, P., Carvalho, G. & Glover, K. A., 28 Sept 2021, In: Evolutionary Applications. 14, 9, p. 2319-2332 14 p.
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental DNA provides higher resolution assessment of riverine biodiversity and ecosystem function via spatio-temporal nestedness and turnover partitioning
Seymour, M., Edwards, F. K., Cosby, B. J., Bista, I., Scarlett, P. M., Brailsford, F., Glanville, H., de Bruyn, M., Carvalho, G. & Creer, S., 3 May 2021, In: Communications Biology. 4, 1, 512.
Research output: Contribution to journal › Article › peer-review - PublishedMolecular characterization of a marine turtle tumor epizootic, profiling external, internal and postsurgical regrowth tumors
Yetsko, K., Farrell, J., Blackburn, N. B., Whitmore, L., Stammnitz, M. R., Whilde, J., Eastman, C., Ramia, D. R., Thomas, R., Krstic, A., Linser, P., Creer, S., Carvalho, G., Devlin, M. A., Nahvi, N., Leandro, A. C., deMaar, T. W., Burkhalter, B., Murchison, E. P., Schnitzler, C. & Duffy, D., 1 Feb 2021, In: Communications Biology. 4, 1, 152.
Research output: Contribution to journal › Article › peer-review
2020
- PublishedComparative genetic stock structure in three species of commercially exploited Indo-Malay Carangidae (Teleosteii, Perciformes)
Jaafar, T. N. A. M., Taylor, M. I., Nor, S. A. M., de Bruyn, M. & Carvalho, G. R., 20 Feb 2020, In: Journal of Fish Biology. 96, 2, p. 337-349
Research output: Contribution to journal › Article › peer-review - PublishedDisentangling the effects of sex, life history and genetic background in Atlantic salmon: growth, heart and liver under common garden conditions
Perry, W. B., Solberg, M. F., Brodie, C., Medina, A. C., Pillay, K. G., Egerton, A., Harvey, A., Creer, S., Llewellyn, M., Taylor, M., Carvalho, G. & Glover, K. A., 7 Oct 2020, In: Royal Society Open Science. 7, 10, 200811.
Research output: Contribution to journal › Article › peer-review - PublishedEcological community dynamics: 20 years of moth sampling reveals the importance of generalists for community stability
Carvalho, G., Seymour, M., Brown, N., Wood, C., Goertz, S. & de Bruyn, M., 1 Dec 2020, In: Basic and Applied Ecology. 49, p. 34-44 10 p.
Research output: Contribution to journal › Article › peer-review - PublishedEpistatic regulation of growth in Atlantic salmon revealed: a QTL study performed on the domesticated-wild interface
Besnier, F., Solberg, M. F., Harvey, A., Carvalho, G., Bekkevold, D., Taylor, M. I., Creer, S., Nielsen, E. E., Skaala, O., Ayllon, F., Dahle, G. & Glover, K. A., 7 Feb 2020, In: BMC Genetics. 21, 1, 13.
Research output: Contribution to journal › Article › peer-review - PublishedExecuting multi-taxa eDNA ecological assessment via traditional metrics and interactive networks
Seymour, M., Edwards, F., Cosby, B., Kelly, M., de Bruyn, M., Carvalho, G. & Creer, S., 10 Aug 2020, In: Science of the Total Environment. 729, 138801.
Research output: Contribution to journal › Article › peer-review - PublishedPhylogenomics and species delimitation for effective conservation of manta and devil rays
Hosegood, J., Humble, E., Ogden, R., De Bruyn, M., Creer, S., Stevens, G. M. W., Abudaya, M., Bassos-Hull, K., Bonfil, R., Fernando, D., Foote, A., Hipperson, H., Jabado, R. W., Kaden, J., Moazzam, M., Peel, L. R., Pollett, S., Ponzo, A., Poortvliet, M., Salah, J., Senn, H., Stewart, J. D., Wintner, S. & Carvalho, G., Dec 2020, In: Molecular Ecology. 29, 24, p. 4783-4796
Research output: Contribution to journal › Article › peer-review
2019
- PublishedDeep segregation in the open ocean: Macaronesia as an evolutionary hotspot for low dispersal marine invertebrates
Vieira, P. E., Desiderato, A., Holdich, D. M., Soares, P., Creer, S., Carvalho, G., Costa, F. O. & Queiroga, H., 1 Apr 2019, In: Molecular Ecology. 28, 7, p. 1784-1800
Research output: Contribution to journal › Article › peer-review - PublishedDemographic reconstruction from ancient DNA supports rapid extinction of the great auk
Thomas, J., Carvalho, G., Haile, J., Rawlence, N. J., Martin, M. D., Ho, S. YW., Sigfússon, A. Þ., Jósefsson, V. A., Frederiksen, M., Linnebjerg, J. F., Samaniego Castruita, J. A., Niemann, J., Sinding, M-H. S., Sandoval-Velasco, M., Soares, A. ER., Lacy, R., Barilaro, C., Best, J., Brandis, D., Cavallo, C., Elorza, M., Garrett, K. L., Groot, M., Johansson, F., Lifjeld, J. T., Nilson, G., Serjeanston, D., Sweet, P., Fuller, E., Hufthammer, A. K., Meldgaard, M., Fjeldsa, J., Shapiro, B., Hofreiter, M., Stewart, J. R., Gilbert, M. T. P. & Knapp, M., 26 Nov 2019, In: Elife. 8, e47509.
Research output: Contribution to journal › Article › peer-review - PublishedDetection of introduced and resident marine species using environmental DNA metabarcoding of sediment and water
Holman, L. E., de Bruyn, M., Creer, S., Carvalho, G., Robidart, J. & Rius, M., 9 Aug 2019, In: Scientific Reports. 9, 11559.
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental DNA size sorting and degradation experiment indicates the state of Daphnia magna mitochondrial and nuclear eDNA is subcellular
Moushomi, R., Wilgar, G., Carvalho, G., Creer, S. & Seymour, M., 29 Aug 2019, In: Scientific Reports. 9, 12500 (2019.
Research output: Contribution to journal › Article › peer-review - PublishedEvolutionary drivers of kype size in Atlantic salmon (Salmo salar): domestication, age and genetics
Perry, W. B., Solberg, M. F., Besnier, F., Dyrhovden, L., Matre, I. H., Fjelldal, P. G., Ayllon, F., Creer, S., Llewelyn, M., Taylor, M. I., Carvalho, G. & Glover, K. A., 17 Apr 2019, In: Royal Society Open Science. 6, 4, 190021.
Research output: Contribution to journal › Article › peer-review
2018
- PublishedAcidity promotes degradation of multi-species environmental DNA in lotic mesocosms
Seymour, M., Durance, I., Cosby, B. J., Ransom-Jones, E., Deiner, K., Ormerod, S. J., Colbourne, J. K., Wilgar, G., Carvalho, G., De Bruyn, M., Edwards, F., Emmett, B. A., Bik, H. M. & Creer, S., 22 Jan 2018, In: Communications Biology. 1, 4.
Research output: Contribution to journal › Article › peer-review - PublishedPerformance of amplicon and shotgun sequencing for accurate biomass estimation in invertebrate community samples
Bista, I., Carvalho, G., Tang, M., Walsh, K., Zhou, X., Hajibabaei, M., Shokralla, S., Seymour, M., Bradley, D., Liu, S., Christmas, M. & Creer, S., Sept 2018, In: Molecular Ecology Resources. 18, 5, p. 1020-1034
Research output: Contribution to journal › Article › peer-review - PublishedStepping stones to isolation: Impacts of a changing climate on the connectivity of fragmented fish populations
Young, E. F., Tysklind, N., Meredith, M. P., de Bruyn, M., Belchier, M., Murphy, E. J. & Carvalho, G. R., Jul 2018, In: Evolutionary Applications. 11, 6, p. 978-994
Research output: Contribution to journal › Article › peer-review - PublishedWhole genome duplication and transposable element proliferation drive genome expansion in Corydoradinae catfishes
Marburger, S., Alexandrou, M., Taggart, J. B., Creer, S., Carvalho, G., Oliveira, C. & Taylor, M. I., 14 Feb 2018, In: Proceedings of the Royal Society B: Biological Sciences. 285, 1872, 20172732.
Research output: Contribution to journal › Article › peer-review
2017
- PublishedAnnual time-series analysis of aqueous eDNA reveals ecologically relevant dynamics of lake ecosystem biodiversity
Bista, I., Carvalho, G., Walsh, K., Seymour, M., Hajibabaei, M., Lallias, D., Christmas, M. & Creer, S., Jan 2017, In: Nature Communications. 8, 14087.
Research output: Contribution to journal › Article › peer-review - PublishedParasitism perturbs the mucosal microbiome of Atlantic Salmon
Llewellyn, M. S., Leadbeater, S., Garcia, C., Sylvain, F-E., Custodio, M., Ang, K. P., Powell, F., Carvalho, G., Creer, S., Elliot, J. & Derome, N., 2017, In: Scientific Reports. 7, 43465.
Research output: Contribution to journal › Article › peer-review - PublishedPlio-Pleistocene phylogeography of the Southeast Asian blue panchax killifish, Aplocheilus panchax.
Beck, S., Carvalho, G., Barlow, A., Ruber, L., Tan, H. H., Nugroho, E., Wowor, D., Nor, S. A. M., Herder, F., Muchlisin, Z. A. & De Bruyn, M., 25 Jul 2017, In: PLoS ONE.
Research output: Contribution to journal › Article › peer-review
2016
- PublishedA common garden design reveals population-specific variability in potential impacts of hybridization between populations of farmed and wild Atlantic salmon, Salmo salar L.
Harvey, A., Glover, K. A., Taylor, M., Creer, S. & Carvalho, G., 1 Mar 2016, In: Evolutionary Applications. 9, 3, p. 435-449
Research output: Contribution to journal › Article › peer-review - PublishedDoes density influence relative growth performance of farmed, wild, and F1 hybrid Atlantic salmon in semi-natural and hatchery common garden conditions?
Harvey, A., Carvalho, G., Taylor, M., Solberg, M. F., Creer, S., Dyrhovden, L., Matre, I. H. & Glover, K. A., 6 Jul 2016, In: Royal Society Open Science. 3, 7, 160152.
Research output: Contribution to journal › Article › peer-review - PublishedEvaluating genetic traceability methods for captive-bred marine fish and their applications in fisheries management and wildlife forensics
Bylemans, J., Maes, G. E., Diopere, E., Cariani, A., Senn, H., Taylor, M. I., Helyar, S., Bargelloni, L., Bonaldo, A., Carvalho, G., Guarniero, I., Komen, H., Martinsohn, J. T., Nielsen, E. E., Tinti, F., Volckaert, F. A. M. & Ogden, R., 25 Feb 2016, In: Aquaculture Environment Interactions. 8, p. 131-145
Research output: Contribution to journal › Article › peer-review - PublishedFish, genes and genomes: contributions to ecology, evolution and management
Carvalho, G., Hauser, L., Martinsohn, J. & Naish, K., 5 Dec 2016, In: Journal of Fish Biology. 89, 6, p. 2471-2478
Research output: Contribution to journal › Special issue › peer-review - PublishedNo loss of genetic diversity in the exploited and recently collapsed population of Bay of Biscay anchovy (Engraulis encrasicolus, L.)
Montes, I., Irondo, M., Manzano, C., Santos, M., Conklin, D., Carvalho, G., Irigoien, X. & Estonba, A., May 2016, In: Marine Biology. 163, 98.
Research output: Contribution to journal › Article › peer-review - PublishedPlasticity in growth of farmed and wild Atlantic salmon: is the increased growth rate of farmed salmon caused by evolutionary adaptations to the commercial diet?
Harvey, A., Solberg, M. F., Troianou, E., Carvalho, G., Taylor, M. I., Creer, S., Dyrhovden, L., Matre, I. H. & Glover, K. A., 1 Dec 2016, In: BMC Evolutionary Biology. 2016, 16, 264.
Research output: Contribution to journal › Article › peer-review - PublishedPlasticity in response to feed availability: Does feeding regime influence the relative growth performance of domesticated, wild and hybrid Atlantic salmon Salmo salar parr?
Harvey, A., Solberg, M. F., Glover, K. A., Taylor, M. I., Creer, S. & Carvalho, G., 1 Sept 2016, In: Journal of Fish Biology. 89, 3, p. 1754-1768
Research output: Contribution to journal › Article › peer-review - PublishedThe biogeography of the atlantic salmon (Salmo salar) gut microbiome
Llewellyn, M. S., McGinnity, P., Dionne, M., Letourneau, J., Thonier, F., Carvalho, G., Creer, S. & Derome, N., May 2016, In: ISME Journal. 10, p. 1280-1284
Research output: Contribution to journal › Article › peer-review - PublishedWorldwide Analysis of Sedimentary DNA Reveals Major Gaps in Taxonomic Knowledge of Deep-Sea Benthos
Sinniger, F., Pawlowski, J., Harii, S., Gooday, A. J., Yamamoto, H., Chevaldonne, P., Cedhagen, T., Carvalho, G. & Creer, S., 14 Jun 2016, In: Frontiers in Marine Science. 3, 92
Research output: Contribution to journal › Article › peer-review
2015
- PublishedGene-associated markers can assign origin in a weakly structured fish, Atlantic herring
Bekkevold, D., Helyar, S. J., Limborg, M. T., Nielsen, E. E., Hemmer-Hansen, J., Clausen, L. A. & Carvalho, G. R., 23 Feb 2015, In: ICES Journal of Marine Science. 72, 6, p. 1790-1810
Research output: Contribution to journal › Article › peer-review - PublishedOceanography and life history predict contrasting genetic population structure in two Antarctic fish species
Young, E. F., Belchier, M., Hauser, L., Horsburgh, G. J., Meredith, M. P., Murphy, E. J., Pascoal, S., Rock, J., Tysklind, N. & Carvalho, G. R., 16 Apr 2015, In: Evolutionary Applications. 8, 5, p. 486-509
Research output: Contribution to journal › Article › peer-review
2014
- PublishedA reliable DNA barcode reference library for the identification of the North European shelf fish fauna
Knebelsberger, T., Landi, M., Neumann, H., Sell, A. F., Campbell, P. D., Laakmann, S., Raupach, M. J., Carvalho, G. R. & Costa, F. O., 12 Mar 2014, In: Molecular Ecology Resources. 14, 5, p. 1060-1071
Research output: Contribution to journal › Article › peer-review - PublishedBorneo and Indochina are Major Evolutionary Hotspots for Southeast Asian Biodiversity
De Bruyn, M., de Bruyn, M., Stelbrink, B., Morley, R. J., Hall, R., Carvalho, G. R., Cannon, C. H., van den Bergh, G., Meijaard, E., Metcalfe, I., Boitani, L., Maiorano, L. & Shoup, R. V., 30 Nov 2014, In: Systematic Biology. 63, 6, p. 879-901
Research output: Contribution to journal › Article › peer-review - PublishedDNA barcoding for species assignment: the case of Mediterranean marine fishes
Carvalho, G. R., Landi, M. D., Arculeo, M., Biondo, G., Martins, R. & Carneiro, M., 15 Sept 2014, In: PLoS ONE. 9, 9
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental DNA for Wildlife Biology and Biodiversity Monitoring
Evans, A. R., Bohmann, K., Evans, A., Gilbert, M. T., Carvalho, G. R., Creer, S., Knapp, M., Yu, D. W. & De Bruyn, M., 10 May 2014, In: Trends in Ecology and Evolution. 29, 6, p. 358-367
Research output: Contribution to journal › Article › peer-review - PublishedFish Product Mislabelling: Failings of Traceability in the Production Chain and Implications for Illegal, Unreported and Unregulated (IUU) Fishing
De Bruyn, M., Helyar, S. J., Lloyd, H., de Bruyn, M., Leake, J., Bennett, N. & Carvalho, G. R., 12 Jun 2014, In: PLoS ONE. 9, 6, p. e98691
Research output: Contribution to journal › Article › peer-review - PublishedMetagenetic analysis of patterns of distribution and diversity of marine meiobenthic eukaryotes
Fonseca, V. G., Carvalho, G. R., Nichols, B., Quince, C., Johnson, H. F., Neill, S. P., Lambshead, J. D., Thomas, W. K., Power, D. M. & Creer, S., 25 Aug 2014, In: Global Ecology and Biogeography. 23, 11, p. 1293–1302
Research output: Contribution to journal › Article › peer-review
2013
- PublishedA genomic island linked to ecotype divergence in Atlantic cod
Hemmer-Hansen, J., Nielsen, E. E., Therkildsen, N. O., Taylor, M. I., Ogden, R., Geffen, A. J., Bekkevold, D., Helyar, S., Pampoulie, C., Johansen, T., FishPopTrace Consortium, N. V. & Carvalho, G. R., 1 May 2013, In: Molecular Ecology. 22, 10, p. 2653-2667
Research output: Contribution to journal › Article › peer-review - PublishedCombination of genetics and spatial modelling highlights the sensitivity of cod (Gadus morhua) population diversity in the North Sea to distributions of fishing
Carvalho, G. R., Heath, M. R., Culling, M. A., Crozier, W. W., Fox, C. J., Gurney, W. S., Hutchinson, W. F., Nielsen, E. E., O'Sullivan, M., Preedy, K. F., Righton, D. A., Speirs, D. C., Taylor, M. I. & Wright, P. J., 17 Nov 2013, In: ICES Journal of Marine Science. 71, 4, p. 794-807
Research output: Contribution to journal › Article › peer-review - PublishedExperimental harvesting of fish populations drives genetically based shifts in body size and maturation
Van Wijk, S. J., van Wijk, S. J., Taylor, M. I., Creer, S., Dreyer, C., Rodrigues, F. M., Ramnarine, I. W., van Oosterhout, C. & Carvalho, G. R., 1 May 2013, In: Frontiers in Ecology and the Environment. 11, 4, p. 181–187
Research output: Contribution to journal › Article › peer-review - PublishedInvestigating the molecular systematic relationships amongst selected Plesionika (Decapoda: Pandalidae) from the Northeast Atlantic and Mediterranean Sea
da Silva, J. M., dos Santos, A., Cunha, M. R., Costa, F. O., Creer, S. & Carvalho, G. R., 1 Jun 2013, In: Marine Ecology. 34, 2, p. 157-170
Research output: Contribution to journal › Article › peer-review - PublishedOutlier SNP markers reveal fine-scale genetic structuring across European hake populations (Merluccius merluccius)
Milano, I., Babbucci, M., Cariani, A., Atanassova, M., Bekkevold, D., Carvalho, G., Espineira, M., Fiorentino, F., Garofalo, G., Geffen, A., Hansen, J. H., Helyar, S. J., Nielsen, E. E., Ogden, R., Patarnello, T., Stagioni, M., Tinti, F. & Bargelloni, L., 18 Nov 2013, In: Molecular Ecology. 23, 1, p. 118-135
Research output: Contribution to journal › Article › peer-review - PublishedPaleo-Drainage Basin Connectivity Predicts Evolutionary Relationships across Three Southeast Asian Biodiversity Hotspots
De Bruyn, M., de Bruyn, M., Rüber, L., Nylinder, S., Stelbrink, B., Lovejoy, N. R., Lavoué, S., Tan, H. H., Nugroho, E., Wowor, D., Ng, P. K., Azizah, M. N., Von Rintelen, T., Hall, R. & Carvalho, G. R., 1 May 2013, In: Systematic Biology. 62, 3, p. 398-410
Research output: Contribution to journal › Article › peer-review - PublishedPopulation genetics provides new insights into biomarker prevalence in dab (Limanda limanda L.): a key marine biomonitoring species
Tysklind, N., Taylor, M. I., Lyons, B. P., Goodsir, F., McCarthy, I. D. & Carvalho, G. R., 1 Sept 2013, In: Evolutionary Applications. 6, 6, p. 891-909
Research output: Contribution to journal › Article › peer-review - PublishedSNP Discovery in European Anchovy (Engraulis encrasicolus, L) by High-Throughput Transcriptome and Genome Sequencing
Montes, I., Conklin, D., Albania, A., Creer, S., Carvalho, G. R., Santos, M. & Estonba, A., 1 Aug 2013, In: PLoS ONE. 8, 8, p. e70051
Research output: Contribution to journal › Article › peer-review - PublishedTime and space in biogeography: response to Parenti & Ebach
De Bruyn, M., de Bruyn, M., Stelbrink, B., Page, T. J., Phillips, M. J., Lohman, D. J., Albrecht, C., Hall, R., von Rintelen, K., Ng, P. K., Shih, H. T., Carvalho, G. R. & von Rintelen, T., 1 Nov 2013, In: Journal of Biogeography. 40, 11, p. 2204-2206
Research output: Contribution to journal › Article › peer-review - PublishedTranscriptomics and in vivo tests reveal novel mechanisms underlying endocrine disruption in an ecological sentinel, Nucella lapillus
Hughes, R. N., Pascoal, S., Carvalho, G. R., Vasieva, O., Hughes, R., Cossins, A., Fang, Y., Ashelford, K., Olohan, L., Barroso, C., Mendo, S. & Creer, S., 1 Mar 2013, In: Molecular Ecology. 22, 6, p. 1589-1608
Research output: Contribution to journal › Article › peer-review
2012
- PublishedA Ranking System for Reference Libraries of DNA Barcodes: Application to Marine Fish Species from Portugal
Costa, F. O., Landi, M., Martins, R., Costa, M. A., Carneiro, M., Alves, M. J., Steinke, D. & Carvalho, G. R., 25 Apr 2012, In: PLoS ONE. 7, 4, p. e35858
Research output: Contribution to journal › Article › peer-review - PublishedDNA Barcoding Reveals Cryptic Diversity within Commercially Exploited Indo-Malay Carangidae (Teleosteii: Perciformes)
De Bruyn, M., Jaafar, T. N., Taylor, M. I., Nor, S. A., de Bruyn, M. & Carvalho, G. R., 29 Nov 2012, In: PLoS ONE. 7, 11, p. e49623
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental selection on transcriptome-derived SNPs in a high gene flow marine fish, the Atlantic herring (Clupea harengus)
De Bruyn, M., Limborg, M. T., Helyar, S. J., DeBruyn, M., Taylor, M. I., Nielson, E. E., Ogden, R., Carvalho, G. R. & Bekkevold, D., 1 Aug 2012, In: Molecular Ecology. 21, 15, p. 3686-3703
Research output: Contribution to journal › Article › peer-review - PublishedGene-associated markers provide tools for tackling illegal fishing and false eco-certification
Taylor, M. I., Nielsen, E. E., Cariani, A., Aoidh, E. M., Maes, G. E., Milano, I., Ogden, R., Taylor, M., Hemmer-Hansen, J., Babbucci, M., Bargelloni, L., Bekkevold, D., Diopere, E., Grenfell, L., Helyar, S., Limborg, M. T., Martinsohn, J. T., McEwing, R., Panitz, F., Patarnello, T., Tinti, F., Van Houdt, J. K., Volckaert, F. A., Waples, R. S., FishPopTrace consortium, N. V. & Carvalho, G. R., 22 May 2012, In: Nature Communications. 3, p. Article No. 851
Research output: Contribution to journal › Article › peer-review - PublishedMolecular biogeography and phylogeography of the freshwater fauna of the Indo-Australian Archipelago
De Bruyn, M., de Bruyn, M., von Rintelen, K., von Rintelen, T., Mather, P. B. & Carvalho, G. R., 1 Jul 2012, Biotic Evolution and Environmental Change in Southeast Asia. 2012 ed. Cambridge University Press
Research output: Chapter in Book/Report/Conference proceeding › Chapter - PublishedPhysical and behavioural influences on larval fish retention: contrasting patterns in two Antarctic fishes
Young, E. F., Rock, J., Meredith, M. P., Belchier, M., Murphy, E. J. & Carvalho, G. R., 28 Sept 2012, In: Marine Ecology Progress Series. 465, p. 201-215
Research output: Contribution to journal › Article › peer-review - PublishedPlastic and Heritable Components of Phenotypic Variation in Nucella lapillus: An Assessment Using Reciprocal Transplant and Common Garden Experiments
Hughes, R. N., Pascoal, S., Carvalho, G. R., Creer, S., Rock, J., Kawaii, K., Mendo, S. & Hughes, R., 27 Jan 2012, In: PLoS ONE. 7, 1, p. e30289
Research output: Contribution to journal › Article › peer-review - PublishedSNP Discovery Using Next Generation Transcriptomic Sequencing in Atlantic Herring (Clupea harengus)
Helyar, S. J., Limborg, M. T., Bekkevold, D., Babbucci, M., van Houdt, J., Maws, G. E., Bargelloni, L., Nielsen, R. O., Taylor, M. I., Ogden, R., Cariani, A., Carvalho, G. R., FishPopTrace Consortium, N. V. & Panitz, F., 7 Aug 2012, In: PLoS ONE. 7, 8, p. e42089
Research output: Contribution to journal › Article › peer-review - PublishedSample richness and genetic diversity as drivers of chimera formation in nSSU metagenetic analyses
Fonseca, V. G., Nichols, B., Lallias, D., Quince, C., Carvalho, G. R., Power, D. M. & Creer, S., 25 Jan 2012, In: Nucleic Acids Research. 40, 9, p. e66
Research output: Contribution to journal › Article › peer-review - PublishedSpecies, sex, size and male maturity composition of previously unreported elasmobranch landings in Kuwait, Qatar and Abu Dhabi Emirate
Moore, A. B., McCarthy, I. D., Carvalho, G. R. & Pierce, R., 1 Apr 2012, In: Journal of Fish Biology. 80, 5, p. 1619-1642
Research output: Contribution to journal › Article › peer-review
2011
- PublishedAnonymous nuclear markers for halfbeak fishes of the genus Hemirhamphodon
De Bruyn, M., Carvalho, G. R., de Bruyn, M. & Grail, W. C., 1 Jan 2011, In: Conservation Genetics Resources. 3, 1, p. 155-157
Research output: Contribution to journal › Article › peer-review - PublishedAnonymous nuclear markers for the Blue Panchax killifish (Aplocheilus panchax)
De Bruyn, M., de Bruyn, M., Grail, W. & Carvalho, G. R., 1 Jan 2011, In: Conservation Genetics Resources. 3, 1, p. 53-55
Research output: Contribution to journal › Article › peer-review - PublishedApplication of SNPs for population genetics of nonmodel organisms: new opportunities and challenges
Helyar, S., Hemmer-Hansen, J., Bekkevold, D., Taylor, M. I., Ogden, R., Limborg, M. T., Cariani, A., Maes, G. E., Diopere, E., Carvalho, G. & Nielsen, A., Mar 2011, In: Molecular Ecology Resources. 11, S1, p. 123-136
Research output: Contribution to journal › Article › peer-review - PublishedBiogeography of the Indo-Australian Archipelago
De Bruyn, M., Carvalho, G. R., Lohman, D. J., de Bruyn, M., Page, T., von Rintelen, K., Hall, R., Ng, P. K., Shih, H. T. & von Rintelen, T., 1 Dec 2011, In: Annual Review of Ecology, Evolution, and Systematics. 42, p. 205-226
Research output: Contribution to journal › Article › peer-review - PublishedDetecting population structure in a high gene-flow species, Atlantic herring (Clupea harengus): direct, simultaneous evaluation of neutral vs putatively selected loci.
Andre, C., Larsson, L. C., Laikre, L., Bekkevold, D., Brigham, J., Carvalho, G. R., Dahlgren, T. G., Hutchinson, W. F., Mariani, S., Mudde, K., Ruzzante, D. E. & Ryman, N., 1 Feb 2011, In: Heredity. 106, 2, p. 270-280
Research output: Contribution to journal › Article › peer-review - PublishedFaunal histories from Holocene ancient DNA
De Bruyn, M., de Bruyn, M., Hoelzel, A. R., Carvalho, G. R. & Hofreiter, M., 1 Aug 2011, In: Trends in Ecology and Evolution. 26, 8, p. 405-413
Research output: Contribution to journal › Article › peer-review - PublishedNovel Tools for Conservation Genomics: Comparing Two High-Throughput Approaches for SNP Discovery in the Transcriptome of the European Hake
Milano, I., Babbucci, M., Panitz, F., Ogden, R., Nielsen, R. O., Taylor, M. I., Helyar, S. J., Carvalho, G. R., Espiñeira, M., Atanassova, M., Tinti, F., Mae, G. E., Patarnello, T. & Bargelloni, L., 22 Nov 2011, In: PLoS ONE. 6, 11, p. e28008
Research output: Contribution to journal › Article › peer-review - PublishedSuccessful extraction of DNA from archived alcohol-fixed white-eye fish specimens using an ancient DNA protocol
De Bruyn, M., DeBruyn, M., Parenti, L. R. & Carvalho, G. R., 1 Jun 2011, In: Journal of Fish Biology. 78, 7, p. 2074-2079
Research output: Contribution to journal › Article › peer-review - PublishedTemporal genetic homogeneity among shore crab (Carcinus maenas) larval events supplied to an estuarine system on the Portuguese northwest coast.
Domingues, C. P., Creer, S., Taylor, M. I., Queiroga, H. & Carvalho, G. R., 1 May 2011, In: Heredity. 106, 5, p. 832-840
Research output: Contribution to journal › Article › peer-review
2010
- PublishedAnonymous nuclear markers for SouthEast Asian halfbeak fishes (Dermogenys)
De Bruyn, M., Grail, W., Barlow, A. & Carvalho, G. R., 1 Sept 2010, In: Conservation Genetics Resources. 2, 1, p. 325-327
Research output: Contribution to journal › Article › peer-review - PublishedGenetic structure of Carcinus maenas within its native range: larval dispersal and oceanographic variability.
Domingues, C. P., Creer, S., Taylor, M. I., Queiroga, H. & Carvalho, G. R., 14 Jul 2010, In: Marine Ecology Progress Series. 410, p. 111-123
Research output: Contribution to journal › Article › peer-review - PublishedHigh-resolution modelling of the shelf and open ocean adjacent to South Georgia, Southern Ocean
Young, E. F., Meredith, M. P., Murphy, E. J. & Carvalho, G. R., 16 Dec 2010, In: Deep Sea Research II: Topical Studies in Oceanography.
Research output: Contribution to journal › Article › peer-review - PublishedMolecular sexing of African rhinoceros.
Peppin, L., McEwing, R., Ogden, R., Hermes, R., Harper, C., Guthrie, A. & Carvalho, G. R., 1 Jun 2010, In: Conservation Genetics. 11, 3, p. 1181-1184
Research output: Contribution to journal › Article › peer-review - PublishedPopulation structure on breeding grounds of Lake Malawi's 'twilight zone' cichlid fishes.
Genner, M. J., Nichols, P., Shaw, P. W., Carvalho, G. R., Robinson, R. L. & Turner, G. F., 1 Feb 2010, In: Journal of Biogeography. 37, 2, p. 258-269
Research output: Contribution to journal › Article › peer-review - PublishedSecond-generation environmental sequencing unmasks marine metazoan biodiversity.
Fonseca, V. G., Carvalho, G. R., Sung, W., Johnson, H. F., Power, D. M., Neill, S. P., Packer, M., Blaxter, M. L., Lambshead, J. D., Thomas, W. K. & Creer, S., 19 Oct 2010, In: Nature Communications. 1, 7, p. 98
Research output: Contribution to journal › Article › peer-review - PublishedUltrasequencing of the meiofaunal biosphere: practice, pitfalls and promises.
Creer, S., Fonseca, V. G., Porazinska, D. L., Giblin-Davis, R. M., Sung, W., Powers, D. M., Packer, M., Carvalho, G. R., Blaxter, M. L., Lambshead, P. J. & Thomas, W. K., 1 Mar 2010, In: Molecular Ecology. 19, 1, p. 4-20
Research output: Contribution to journal › Article › peer-review
2009
- PublishedDevelopment and Application of Microsatellites in Carcinus maenas: Genetic Differentiation between Northern and Central Portuguese Populations
Pascoal, S., Creer, S., Taylor, M. I., Queiroga, H., Carvalho, G. & Mendo, S., 30 Sept 2009, In: PLoS ONE. 4, 9, p. e7268
Research output: Contribution to journal › Article › peer-review - PublishedDevelopment of 30 microsatellite markers for dab (Limanda limanda L.): a key UK marine biomonitoring species.
Tysklind, N., Taylor, M. I., Lyons, B. P., McCarthy, I. D. & Carvalho, G. R., 1 May 2009, In: Molecular Ecology Resources. 9, 3, p. 951-955
Research output: Contribution to journal › Article › peer-review - PublishedDisentangling the effects of evolutionary, demographic, and environmental factors influencing genetic structure of natural populations: Atlantic herring as a case study.
Gaggiotti, O. E., Bekkevold, D., Jorgensen, H. B., Foll, M., Carvalho, G. R., Andre, C. & Ruzzante, D. E., 1 Nov 2009, In: Evolution. 63, 11, p. 2939-2951
Research output: Contribution to journal › Article › peer-review - PublishedGenomic signatures of local directional selection in a high gene flow marine organism; the Atlantic cod (Gadus morhua).
Nielsen, E. E., Hemmer-Hansen, J., Poulsen, N. A., Loeschcke, V., Moen, T., Johansen, T., Mittelholzer, C., Taranger, G. L., Ogden, R. & Carvalho, G. R., 1 Dec 2009, In: BMC Evolutionary Biology. 9, p. 276
Research output: Contribution to journal › Article › peer-review - PublishedIsolation and characterization of 28 new microsatellite markers for European flounder (Platichthys flesus L.)
Tysklind, N., Neuparth, T., Ashcroft, G. R., Taylor, M. I., Lyons, B. P., McCarthy, I. D. & Carvalho, G. R., 1 May 2009, In: Molecular Ecology Resources. 9, 3, p. 1065-1068
Research output: Contribution to journal › Article › peer-review - PublishedPatterns of self compatibility, inbreeding depression, outcrossing, and sex allocation in a marine bryozoan suggest the predominating influence of sperm competition.
Hughes, R. N., Wright, P. J., Carvalho, G. R. & Hutchinson, W. F., 1 Nov 2009, In: Biological Journal of the Linnean Society. 98, 3, p. 519-531
Research output: Contribution to journal › Article › peer-review
2008
- PublishedA DNA-Based Approach for the Forensic Identification of Asiatic Black Bear (Ursus thibetanus) in a Traditional Asian Medicine.
Peppin, L., McEwing, R., Carvalho, G. R. & Ogden, R., 1 Nov 2008, In: Journal of Forensic Sciences. 53, 6, p. 1358-1362
Research output: Contribution to journal › Article › peer-review - PublishedDNA barcodes of fish of the Scotia Sea, Antarctica indicate priority groups for taxonomic and systematics focus.
Rock, J., Costa, F. O., Walker, D. I., North, A. W., Hutchinson, W. F. & Carvalho, G. R., 1 Jun 2008, In: Antarctic Science. 20, 3, p. 253-262
Research output: Contribution to journal › Article › peer-review - PublishedGenetic homogeneity among breeding grounds and nursery areas of an exploited Lake Malawi cichlid fish.
Genner, M. J., Nichols, P., Shaw, P. W., Carvalho, G. R., Robinson, R. L. & Turner, G. F., 1 Sept 2008, In: Freshwater Biology. 53, 9, p. 1823-1831
Research output: Contribution to journal › Article › peer-review - PublishedGenetic markers in biomonitoring programmes: providing a population structure framework for data interpretation.
Tysklind, N., Lyons, B. P., Taylor, M. I., McCarthy, I. D. & Carvalho, G. R., 1 Jan 2008.
Research output: Contribution to conference › Paper - PublishedMolecular phylogeny supports division of the 'cosmopolitan' taxon Celleporella (Bryozoa; Cheilostomata) into four major clades.
Hughes, R. N., Gomez, A., Wright, P. J., Moyano, H. I., Cancino, J. M., Carvalho, G. R. & Lunt, D. H., 1 Jan 2008, In: Molecular Phylogenetics and Evolution. 46, 1, p. 369-374
Research output: Contribution to journal › Article › peer-review - PublishedParadigm shifts in marine fisheries genetics: ugly hypotheses slain by beautiful facts.
Hauser, L. & Carvalho, G. R., 1 Dec 2008, In: Fish and Fisheries. 9, 4, p. 333-362
Research output: Contribution to journal › Article › peer-review - PublishedStrategies for using population genetic markers in the European sentinel flatfish species Dab (Limanda limanda)
Lyons, B. P., Tysklind, N., Stentiford, G. D., Feist, S. W., Taylor, M. I., McCarthy, I. D. & Carvalho, G. R., 1 Jul 2008, In: Marine Environmental Research. 66, 1, p. 179-180
Research output: Contribution to journal › Article › peer-review
2007
- PublishedAge of cichlids: New dates for ancient lake fish radiations
Genner, M. J., Seehausen, O., Lunt, D. H., Joyce, D. A., Shaw, P. W., Carvalho, G. R. & Turner, G. F., 1 May 2007, In: Molecular Biology and Evolution. 24, 5, p. 1269-1282
Research output: Contribution to journal › Article › peer-review - PublishedComparative phylogenography of endemic cyprinids in the south-west Iberian Peninsula: evidence for a new ichthyogeographic area.
Mesquita, N., Cunha, C., Carvalho, G. R. & Coelho, M. M., 1 Aug 2007, In: Journal of Fish Biology. 71, Supplement A, p. 45-75
Research output: Contribution to journal › Article › peer-review - PublishedConcordance of allozyme and microsatellite differentiation in a marine fish, but evidence of selection at a microsatellite locus.
Larsson, L. C., Laikre, L., Palm, S., Andre, C., Carvalho, G. R. & Ryman, N., 1 Mar 2007, In: Molecular Ecology. 16, 6, p. 1135-1147
Research output: Contribution to journal › Article › peer-review - PublishedEvolution of a cichlid fish in a Lake Malawi satellite lake.
Genner, M. J., Nichols, P., Carvalho, G. R., Robinson, R. L., Shaw, P. W., Smith, A. & Turner, G. F., 22 Sept 2007, In: Proceedings of The Royal Society B - Biological Sciences. 274, 1623, p. 2249-2257
Research output: Contribution to journal › Article › peer-review - PublishedEvolutionary implications of pollutants on genetic variability of sentinel bioindicator flatfish
Tysklind, N., Neuparth, T., Lyons, B. P., Taylor, M. I., McCarthy, I. D. & Carvalho, G. R., 1 Jan 2007.
Research output: Contribution to conference › Paper - PublishedMating trials validate the use of DNA barcoding to reveal cryptic speciation of a marine bryozoan taxon.
Gomez, A., Wright, P. J., Lunt, D. H., Cancino, J. M., Carvalho, G. R. & Hughes, R. N., 22 Jan 2007, In: Proceedings of The Royal Society B - Biological Sciences. 274, 1607, p. 199-207
Research output: Contribution to journal › Article › peer-review - PublishedMitochondrial DNA phylogeography and mating compatibility reveal marked genetic structuring and speciation in the NE Atlantic bryozoan Celleporella hyalina
Gomez, A., Hughes, R. N., Wright, P. J., Carvalho, G. R. & Lunt, D. H., 1 May 2007, In: Molecular Ecology. 16, 10, p. 2173-2188
Research output: Contribution to journal › Article › peer-review - PublishedQuality of hatchery-reared juveniles for marine fisheries stock enhancement.
Le Vay, L., Carvalho, G., Quinitio, E. T., Lebata, J. H., Ut, V. N. & Fushimi, H., 22 Aug 2007, In: Aquaculture. 268, 1-4, p. 169-180
Research output: Contribution to journal › Article › peer-review - PublishedReproductive isolation among deep-water cichlid fishes of Lake Malawi differing in monochromatic male breeding dress
Genner, M. J., Nichols, P., Carvalho, G. R., Robinson, R. L., Shaw, P. W. & Turner, G. F., 1 Feb 2007, In: Molecular Ecology. 16, 3, p. 651-662
Research output: Contribution to journal › Article › peer-review - PublishedValidation of the barcoding gene COI for use in forensic genetic species identification
Dawnay, N., Ogden, R., McEwing, R., Carvalho, G. & Thorpe, R. S., 15 Nov 2007, In: Forensic Science International. 173, 1, p. 1-6
Research output: Contribution to journal › Article › peer-review - PublishedYurii Petrovich Altukhov 1936-2006 - Obituary
Carvalho, G. R. & Thorpe, J., 1 Feb 2007, In: Journal of Fish Biology. 70, 2, p. 331-333
Research output: Contribution to journal › Article › peer-review
2006
- PublishedBiocomplexity in a highly migratory pelagic marine fish, Atlantic herring.
Ruzzante, D. E., Mariani, S., Bekkevold, D., Andre, C., Mosegaard, H., Clausen, L. A., Dahlgren, T. G., Hutchinson, W. F., Hatfield, E. M., Torstensen, E., Brigham, J., Simmonds, E. J., Laikre, L., Larsson, L. C., Stet, R. J., Ryman, N. & Carvalho, G. R., 22 Jun 2006, In: Proceedings of The Royal Society B - Biological Sciences. 273, 1593, p. 1459-1464
Research output: Contribution to journal › Article › peer-review - PublishedEvolutionary implications of pollutants on genetic variability of sentinel bioindicator flatfish.
Tysklind, N., Lyons, B. P., Taylor, M. I., McCarthy, I. D. & Carvalho, G. R., 1 Jan 2006.
Research output: Contribution to conference › Paper - PublishedHeterogeneous evolution of microsatellites revealed by reconstruction of recent mutation history in an invasive apomictic snail, Potamopyrgus antipodarum
Weetman, D., Hauser, L. & Carvalho, G. R., 1 May 2006, In: Genetica. 127, 1-3, p. 285-293
Research output: Contribution to journal › Article › peer-review
2005
- PublishedA molecular approach to detect hybridisation between crucian carp (Carassius carassius) and non-indigenous carp species (Carassius spp. and Cyprinus carpio)
Hanfling, B., Bolton, P., Harley, M. & Carvalho, G. R., 1 Mar 2005, In: Freshwater Biology. 50, 3, p. 403-417
Research output: Contribution to journal › Article › peer-review - PublishedEnvironmental correlates of population differentiation in Atlantic herring
Bekkevold, D., Aandre, C., Dahlgren, T. G., Clausen, L. A., Torstensen, E., Mosegaard, H., Carvalho, G. R., Christensen, T. B., Norlinder, E. & Ruzzante, D. E., 1 Dec 2005, In: Evolution. 59, 12, p. 2656-2668
Research output: Contribution to journal › Article › peer-review - PublishedMacro- and micro-geographic variation in pantophysin (Panl) allele frequencies in NE Atlantic cod Gadus morhua
Case, R. A., Hutchinson, W. F., Hauser, L., Van Oosterhout, C. & Carvalho, G. R., 11 Oct 2005, In: Marine Ecology Progress Series. 301, p. 267-278
Research output: Contribution to journal › Article › peer-review - PublishedParadoxical polyembryony? Embryonic cloning in an ancient order of marine bryozoans
Hughes, R. N., D'Amato, M. E., Bishop, J. D., Carvalho, G. R., Craig, S. F., Hansson, L. J., Harley, M. A. & Pemberton, A. J., 22 Jun 2005, In: Biology Letters. 1, 2, p. 178-180
Research output: Contribution to journal › Article › peer-review - PublishedPhylogeography of the cyprinid Squalius aradensis and implications for conservation of the endemic freshwater fauna of southern Portugal
Mesquita, N., Hanfling, B., Carvalho, G. R. & Coelho, M. M., 1 Jun 2005, In: Molecular Ecology. 14, 7, p. 1939-1954
Research output: Contribution to journal › Article › peer-review - PublishedPopulation genetic structure and history of the long-tailed hake, Macruronus magellanicus, in the SW Atlantic as revealed by mtDNA RFLP analysis
Carvalho, G. R. & D'Amato, M. E., 1 Apr 2005, In: ICES Journal of Marine Science. 62, 2, p. 247-255
Research output: Contribution to journal › Article › peer-review - PublishedThe response of epilithic bacteria to different metals regime in two upland streams: assessed by conventional microbiological methods and PCR-DGGE
Mahmoud, H. M., Goulder, R. & Carvalho, G. R., 1 Jul 2005, In: Archiv Fur Hydrobiologie. 163, 3, p. 405-427
Research output: Contribution to journal › Article › peer-review
2004
- PublishedDNA markers indicate that distinct spawning cohorts and aggregations of Patagonian squid, Loligo gahi, do not represent genetically discrete subpopulations
Shaw, P. W., Arkhipkin, A. I., Adcock, G. J., Burnett, W. J., Carvalho, G. R., Scherbich, J. N. & Villegas, P. A., 1 May 2004, In: Marine Biology. 144, 5, p. 961-970
Research output: Contribution to journal › Article › peer-review - PublishedIdentification and biology of Diplotaxodon, Rhamphochromis and Pallidochromis.
Turner, G. F., Robinson, R. L., Shaw, P. W., Carvalho, G. R. & Snoeks, J. (ed.), 1 Jan 2004, The Cichlid Diversity of Lake Malawi/Nyasa/Niassa: Identification: Distribution and Taxonomy. 2004 ed. Cichlid Press, p. 198-251
Research output: Chapter in Book/Report/Conference proceeding › Chapter
2003
- PublishedInbreeding depression and genetic load of sexually selected traits: how the guppy lost its spots.
Van Oosterhout, C., Trigg, R. E., Carvalho, G. R., Magurran, A. E., Hauser, L. & Shaw, P. W., 1 Mar 2003, In: Journal of Evolutionary Biology. 16, 2, p. 273-281
Research output: Contribution to journal › Article › peer-review - PublishedIsolation and characterization of polymorphic microsatellite loci in the endangered Portuguese freshwater fish Squalis aradensis (Cyprinidae)
Mesquita, N., Cunha, C., Hanfling, B., Carvalho, G. R., Ze-Ze, L., Tenreiro, R. & Coelho, M. M., 1 Dec 2003, In: Molecular Ecology Notes. 3, 4, p. 572-574
Research output: Contribution to journal › Article › peer-review - PublishedMicrosatellite analysis of genetic population structure of the endangered cyprinid Anaecypris hispanica in Portugal: implications for conservation.
Salgueiro, P., Carvalho, G. R., Collares-Pereira, M. J. & Coelho, M. M., 1 Jan 2003, In: Biological Conservation. 109, 1, p. 47-56
Research output: Contribution to journal › Article › peer-review - PublishedTemporal analysis of archived samples indicates marked genetic changes in declining North Sea cod.
Hutchinson, W. F., Van Oosterhout, C., Rogers, S. I. & Carvalho, G. R., 1 Oct 2003, In: Proceedings of The Royal Society B - Biological Sciences. 270, 1529, p. 2125-2132
Research output: Contribution to journal › Article › peer-review - PublishedTemporal stability of genetic population structure in the New Zealand snapper, Pagrus auratus, and relationship to coastal currents
Bernal-Ramirez, J. H., Adcock, G. J., Hauser, L., Carvalho, G. R. & Smith, P. J., 1 Mar 2003, In: Marine Biology. 142, 3, p. 567-574
Research output: Contribution to journal › Article › peer-review
2002
- PublishedA technique for the molecular genetic analysis of Daphnia resting eggs from sub-recent lake sediments.
Reid, V. A., Carvalho, G. R., George, D. G. & Griffiths, H. I., 1 May 2002, In: Journal of Paleolimnology. 27, 4, p. 481-486
Research output: Contribution to journal › Article › peer-review - PublishedComparative demographic impacts of 'info-chemicals' and exploitative competition: an empirical test using Daphnia magna.
Mitchell, S. E. & Carvalho, G. R., 1 Mar 2002, In: Freshwater Biology. 47, 3, p. 459-471
Research output: Contribution to journal › Article › peer-review - PublishedExtensive population subdivision of the cuttlefish Sepia officinalis (Mollusca : Cephalopoda) around the Iberian Peninsula indicated by microsatellite DNA variation.
Perez-Losada, M., Guerra, A., Carvalho, G. R., Sanjuan, A. & Shaw, P. W., 1 Dec 2002, In: Heredity. 89, 6, p. 417-424
Research output: Contribution to journal › Article › peer-review - PublishedLate glacial history of the cold-adapted freshwater fish Cottus gobio, revealed by microsatellites.
Hanfling, B., Hellemans, B., Volckaert, F. A. & Carvalho, G. R., 1 Sept 2002, In: Molecular Ecology. 11, 9, p. 1717-1729
Research output: Contribution to journal › Article › peer-review - PublishedLoss of microsatellite diversity and low effective population size in an overexploited population of New Zealand snapper.
Hauser, L., Adcock, G. J., Smith, P. J., Ramirez, J. H. & Carvalho, G. R., 1 Sept 2002, In: Proceedings of the National Academy of Sciences of the USA. 99, 18, p. 11742-11747
Research output: Contribution to journal › Article › peer-review - PublishedMeasuring genetic variation in wild populations: from molecular markers to adaptive traits.
Carvalho, G. R., Van Oosterhout, C., Hauser, L., Magurran, A. E., Hails, R. (ed.), Beringer, J. (ed.) & Godfray, H. C. (ed.), 1 Jan 2002, Genes in the Environment. 2002 ed. Cambridge University Press, p. 91-111
Research output: Chapter in Book/Report/Conference proceeding › Chapter - PublishedMicro- and macrogeographic genetic structure in bryozoans with different larval strategies.
Porter, J. S., Ryland, J. S. & Carvalho, G. R., 24 Jun 2002, In: Journal of Experimental Marine Biology and Ecology. 272, 2, p. 119-130
Research output: Contribution to journal › Article › peer-review - PublishedPelagic cichlid fishes of Lake Malawi/Nyasa: biology, management and conservation.
Turner, G. F., Robinson, R. L., Ngatunga, B. P., Shaw, P. W., Carvalho, G. R. & Cowx, I. G. (ed.), 1 Jan 2002, Management and Ecology of Lake and Reservoir Fisheries.. 2002 ed. Blackwell, p. 353-367
Research output: Chapter in Book/Report/Conference proceeding › Chapter - PublishedReconstruction of microsatellite mutation history reveals a strong and consistent deletion bias in invasive clonal snails, Potamopyrgus antipodarum.
Weetman, D., Hauser, L. & Carvalho, G. R., 1 Oct 2002, In: Genetics. 162, 2, p. 813-822
Research output: Contribution to journal › Article › peer-review - PublishedSpeciation in ancient cryptic species complexes: evidence from the molecular phylogeny of Brachionus plicatilis (Rotifera).
Gomez, A., Serra, M., Carvalho, G. R. & Lunt, D. H., 1 Jul 2002, In: Evolution. 56, 7, p. 1431-1444
Research output: Contribution to journal › Article › peer-review - PublishedThe interplay between colonization history and gene flow in passively dispersing zooplankton: microsatellite analysis of rotifer resting eg banks.
Gomez, A., Adcock, G. J., Lunt, D. H. & Carvalho, G. R., 1 Jan 2002, In: Journal of Evolutionary Biology. 15, 1, p. 158-171
Research output: Contribution to journal › Article › peer-review - PublishedTiming of the population dynamics of bullhead Cottus gobio (Teleostei : Cottidae) during the Pleistocene.
Volckaert, F. A., Hanfling, B., Hellemans, B. & Carvalho, G. R., 1 Nov 2002, In: Journal of Evolutionary Biology. 15, 6, p. 930-944
Research output: Contribution to journal › Article › peer-review - Publishedmt-DNA sequences and possible invasion pathways of the Chinese mitten crab.
Hanfling, B., Carvalho, G. R. & Brandl, R., 9 Aug 2002, In: Marine Ecology Progress Series. 238, p. 307-310
Research output: Contribution to journal › Article › peer-review
2001
- PublishedHaplotype frequency distribution and discriminatory power of two mtDNA fragments in a marine pelagic teleost (Atlantic herring, Clupea harengus)
Hauser, L., Turan, C. & Carvalho, G. R., 1 Dec 2001, In: Heredity. 87, 6, p. 621-630
Research output: Contribution to journal › Article › peer-review - PublishedIsolation and characterization of di- and trinucleotide microsatellites in the freshwater snail Potamopyrgus antipodarum.
Weetman, D., Hauser, L. & Carvalho, G. R., 1 Sept 2001, In: Molecular Ecology Notes. 1, 3, p. 185-187
Research output: Contribution to journal › Article › peer-review - PublishedIsolation and characterization of microsatellites in the bryozoan Crisia denticulata
Craig, S. F., D'Amato, M. E., Harley, M., Bishop, J., Hughes, R. N. & Carvalho, G., 1 Dec 2001, In: Molecular Ecology Notes. 1, 4, p. 281-282
Research output: Contribution to journal › Article › peer-review - PublishedMarked genetic structuring in localised spawning populations of cod Gadus morhua in the North Sea and adjoining waters, as revealed by microsatellites.
Hutchinson, W. F., Carvalho, G. R. & Rogers, S. I., 28 Nov 2001, In: Marine Ecology Progress Series. 223, p. 251-260
Research output: Contribution to journal › Article › peer-review - PublishedMorphological and genetic adaptation to a lagoon environment: a case study in the bryozoan genus Alcyonidium
Porter, J. S., Dyrynda, P. E., Ryland, J. S. & Carvalho, G. R., 1 Sept 2001, In: Marine Biology. 139, 3, p. 575-585
Research output: Contribution to journal › Article › peer-review - PublishedPelagic cichlid fishes of Lake Malawi/Nyasa.
Turner, G. F., Robinson, R. L., Ngatunga, B. P., Shaw, P. W., Carvalho, G. R. & Coleman, R. M. (ed.), 1 Jan 2001, Cichlid Research: State of the Art. 2001 ed. University of Michigan, p. 287-302
Research output: Chapter in Book/Report/Conference proceeding › Chapter - PublishedRiver basin-related genetic structuring in an endangered fish species, Chondrostoma lusitanicum, based on mtDNA sequencing and RFLP analysis
Mesquita, N., Carvalho, G. R., Shaw, P., Crespo, E. & Coelho, M. M., 1 Mar 2001, In: Heredity. 86, 3, p. 253-264
Research output: Contribution to journal › Article › peer-review
2000
- PublishedGenetic population structure indicates sympatric speciation of Lake Malawi pelagic cichlids.
Shaw, P. W., Turner, G. F., Idid, M. R., Robinson, R. L. & Carvalho, G. R., 22 Nov 2000, In: Proceedings of The Royal Society B - Biological Sciences. 267, 1459, p. 2273-2280
Research output: Contribution to journal › Article › peer-review - PublishedMolecular genetic analysis of Daphnia in the English Lake District: species identity, hybridisation and resting egg banks.
Reid, V. A., Carvalho, G. R. & George, D. G., 1 Jun 2000, In: Freshwater Biology. 44, 2, p. 247-253
Research output: Contribution to journal › Article › peer-review - PublishedPhylogeography and regional endemism of a passively dispersing zooplankter: mitochondrial DNA variation in rotiferresting egg banks.
Gomez, A., Carvalho, G. R. & Lunt, D. H., 7 Nov 2000, In: Proceedings of The Royal Society B - Biological Sciences. 267, 1458, p. 2189-2197
Research output: Contribution to journal › Article › peer-review - PublishedScreening of DNA polymorphisms in samples of archived scales from New Zealand snapper.
Adcock, G. J., Ramirez, J. H., Hauser, L., Smith, P. & Carvalho, G. R., 1 May 2000, In: Journal of Fish Biology. 56, 5, p. 1283-1287
Research output: Contribution to journal › Article › peer-review - PublishedSex, parthenogenesis and genetic structure of rotifers: microsatellite analysis of contemporary and resting egg bank populations.
Gomez, A. & Carvalho, G. R., 1 Feb 2000, In: Molecular Ecology. 9, 2, p. 203-214
Research output: Contribution to journal › Article › peer-review
Past Students and Postdocs/Fellows
Since first appointment to academia in 1990, I have invested considerable effort in the promotion and support of early-career postgraduates and postdoctoral staff, a significant proportion of who are now leaders in Molecular Ecology and Evolution. An early opportunity for such training on a global scale was provided by the award of a NATO Advanced Study Institute ASI) held in Erice, Sicily, together with the resulting textbook: Advances in Molecular Ecology (1998), (Ed. GR Carvalho). The ASI provided the first international opportunity for consideration of advances in concepts, analysis and use of PCR-based tools and in ecology and evolution, with 74 NATO students and 15 lecturers for 10 days of seminars, student poster sessions, debate and hands-on use of new software). Lecturers incl. P Beerli, A Estoup, PDN Hebert, DM Hillis, JB Mitton, LH Riesberg, F Rollo, RC Vrijenhoek, RD Ward, JP Young- individuals that represent pioneers in the field of evolutionary/molecular ecology. ASI students included early career scientists who are now major contributors in molecular ecology, incl. J-C Simon, C Primmer, JC Vogel, B Angers, C Biermann L Bargelloni, A Gómez, K Naish, L Hauser, S Piertney.
Large, interactive research groups were established at Swansea, Hull and Bangor by Carvalho (e.g. Hull (e.g. 1996- Carvalho established a new molecular ecology group, which by 2002 had membership of 35, inc. O. Seehausen, Univ. Berne, T Kocher (Visiting Fellow, now Univ. Maryland), GF Turner (Past Head of Biological Sciences, Bangor University; C. van Oosterhout (now, Reader Univ. East Anglia); A Gómez, (now NERC Advanced Fellow); S. Mariani, (now Professor of Conservation Genetics, Salford Univ.) and Bangor Universities, currently with 4 postdoctoral staff (incl. NERC Fellow), 11 PhD students and 3 Masters students ). Collectively, Carvalho has supervised and collaborated with numerous early-stage scientists (to date: 16 Masters; 36 PhD (24 as lead supervisor); 17 postdoctoral, including 3 Marie-Curie, 1 NATO, 1 United Nations, 3 NERC Fellows, 2 Visiting Professorial Fellowships (TD Kocher, USA; P Bloomer, S Africa), and 9 Erasmus Scholars.
To support early career scientists, the first author of most primary papers is the respective PhD student or postdoctoral scientist. Additionally, where other junior colleagues have made significant input, they are given prominence.
He has acted as external examiner of 35 PhD students, including candidates from James Cook University of N Queensland, Rhodes University, SA, Griffith University, Australia, Guelph University, Canada, Open University (Naples), Stockholm University, University of Cork, University of Dublin, University of Essex, Nottingham University, Bristol University, York University, Bangor University, Plymouth University, University of Leeds, Liverpool John Moores University, University of Stirling, University of Hull, University of Southampton.
A selection of past Carvalho PhD students and postdoctoral Associates/Research Fellows include: KA Naish (PhD student) – Tenured Professor in Molecular Ecology, School of Aquatic Sciences & Fisheries, University of Washington, Seattle; SB Piertney (PhD student) – Professor of Molecular Ecology and Evolution, Aberdeen University, UK; L Hauser (PhD student, PDR) – Tenured Professor in Molecular Ecology, School of Aquatic Sciences & Fisheries, University of Washington, Seattle; R. Idid (PhD student) – Senior Lecturer in Evolutionary Biology, University of Malaya, Kuala Lumpur, Malaysia; C Turan (PhD student) – Professor of Fisheries, Head of Department of Basic Sciences, and past Dean of Faculty of Fisheries, Mustafa Kemal University, Turkey; J Porter (PhD student) – Senior Lecturer in Marine Biology, Heriot Watt University, Edinburgh, UK; H Abdullah (PhD student) – Senior Lecturer in Ecology, Kuwait University; PW Shaw (PDR) –Professor of Population Genetics & Genomics, Leader of Aquatic, Behavioural and Evolutionary Biology (ABEB), Aberystwyth University, UK; A Gómez (PDR) – NERC Advanced Fellow, Hull University, UK; E D’Amato (PDR) – Research Manager, Forensic DNA laboratory, University of the Western Cape, SA; S. Helyar (PDR) – Head of Genetics, MATIS Icelandic Food and & Biotech. Lab R&D, Iceland; C van Ooosterhout (PDR) – Professor of Evolutionary Biology, Univ. East Anglia, UK; J Rock (PDR) – Lecturer, Univ. Otago, New Zealand; MI Taylor (UK-RUC-Fellow) – Senior Lecturer, Univ. East Anglia, UK; S Creer (Fellow) – Senior Lecturer, Bangor University, UK; S Mariani (PDR) –Professor of Conservation Genetics, University Salford, UK; F Costa (Marie-Curie Fellow) – Asst. Professor & Chair, CBOL European FishBol, Univ. Braga, Portugal; M de Bruyn (Fellow/PDR) – Senior Lecturer in Molecular Ecology, Bangor University; 2012 winner of 14th R.J.H. Hintelmann Scientific Award for Zoological Systematics; V. Fonseca (PhD student) – Head of Env. Genomics Section, Zoological Research Museum Alexander Koenig (ZFMK) Germany; N Tysklind (PhD student/PDR) – INRA, Team Leader – Population Genetics and Genomics, French Guyana.



